Key Takeaways:
- Cannabis law and regulations:
- The Narcotic Drugs Amendment Bill, Narcotic Drugs Regulation 2016, Narcotic Drugs Amendment (Cannabis) Regulations 2018 and the Narcotic Drugs Amendment (Medical Cannabis) Bill 2021 regulate the medical cannabis market in Australia.
- In the Australian Capital Territory, Drugs of Dependence (Personal Cannabis Use) Amendment Bill 2018 regulates recreational cannabis in that territory.
- Licenses and permits available:
- Single Medical Cannabis License: Cultivation, production and manufacture of cannabis for medicinal or scientific purposes
- Permits: Cultivation and production; manufacture
- License to import narcotic and psychotropic drugs and precursor substances
- License to export narcotic, psychotropic and precursor substances
- Permit to import narcotic and psychotropic substances to Australia
- Permit to export narcotic and psychotropic substances to Australia
- License or permit to import medicinal cannabis for SAS and AP schemes only.
- Taxes and fees: The goods and services tax (GST) of 10% applies on sale of medical cannabis in Australia. There is no additional tax charged on medical cannabis.
- Market:
- There are two ways medicinal cannabis can be accessed by patients in Australia: through the Special Access Scheme (SAS) and the Authorized Prescriber (AP) pathway. Since its legalization in 2016, more than 3.6 million prescriptions for medical cannabis have been issued in Australia.
- Spending by Australians on medicinal cannabis surpassed $402 million in the first half of 2024. The number of units of medicinal cannabis sold in the first half of 2024 is almost four times the number of units sold in the first half of 2022.
- Australia mainly exports cannabis to New Zealand, UK, and Germany.
- As of December 2025, according to Australia’s Department of Health and Aged Care, there were 39 approved medicinal cannabis cultivators and producers, of which 38 were allowed to cultivate and produce, and 22 were allowed to manufacture. There were 23 licensed medicinal cannabis product manufacturers and 36 licensed importers.
- Australia’s legal cannabis market is forecast to grow from around US$600 million in 2024 to US$1.2 billion in 2028, making it the 5th largest in the world.
Australia Cannabis Legalization
In June 2015, the Lambert family donated $33.7 million to the University of Sydney, establishing the ‘Lambert Initiative’. This is the largest Australian medicinal cannabis research group, providing an invaluable boost to the development of treatments for childhood epilepsy, cancer, chronic pain, obesity, anorexia and dementia.
| Population (m) | 27.74 | |
| GDP (US$ t) | 1.83 | |
| Total Healthcare Expenditure (US$ b, est.) | 182 | |
| Alcohol Consumption (Litres Per Capita Per Year) | 10.6 | |
| Prevalence of Cannabis Users (%) | 11.5 | |
| Source: World Bank/ ABS/ WHO/ DATA UNODC/ Prohibition Partners/ AIHW | ||
A 2016 University of Sydney report, Medicinal Cannabis in Australia: Science, Regulation & Industry, found that the Australian medicinal cannabis market, once opened up, could spark initial demand for as much as 8,000 kg of product.
The Narcotic Drugs Amendment Bill was passed by the Australian Federal Government in February 2016, legalizing medical cannabis in the country and establishing a licensing scheme for its cultivation and manufacture. In October 2016, the Narcotic Drugs Regulation 2016 was published, which added more details to the country’s medical cannabis licensing program. Several States and Territories have since amended and/or introduced new legislation and regulation.
Generally, therapeutic goods must be registered on the Australian Register of Therapeutic Goods (ARTG) to be lawfully supplied in Australia. There are currently only to cannabis-based medicines registered on the ARTG: Sativex and Epidyolex. However, the Therapeutic Goods Administration (TGA) recognises that there are cases where patients need therapeutic goods that are not listed in the ARTG. Therefore, there are two ways that ‘unapproved’ medicinal cannabis can be accessed by patients in Australia: through the Special Access Scheme (SAS) and the Authorised Prescriber (AP) pathway.
| Pathway | How it Works | Reporting/Approval Level |
| Authorised Prescriber | A medical practitioner is granted approval to prescribe a specific category of cannabis for a particular indication (usage) for a class of patients in their immediate care. | Prescriber-level: The medical practitioner must submit patient numbers every 6 months. |
| SAS-B | A registered health practitioner must apply to the TGA for permission for each individual patient. As approval letter must be obtained from the TGA before the product is supplied to the patient. | Patient-level: Each application is unique to one patient and requires clinical justification. |
| SAS-A | A notification-based pathway for patients who are “seriously ill” (defined as having a condition from which death is reasonably likely within months or from which premature death is reasonably likely to occur in the absence of early treatment). | Notification-only: No prior TGA approval is needed, but the medical practitioner must notify the TGA after prescribing. |
| Registered Medicines (ARTG) | Products like Sativex or Epidyolex that have been fully tested and registered. | Prescription-level: No TGA application needed; they are treated like any other standard prescription medicine. |
In January 2018, Australia said it planned to become the fourth country in the world to legalize medicinal cannabis exports in a bid to score a piece of the estimated $57 billion global market. The export of medicinal cannabis products was legalized in February 2018 through the Narcotic Drugs Amendment (Cannabis) Regulations 2018.
Provided domestic supply of medicinal cannabis is not affected, the following products are eligible for export if granted a license and permit to export:
- Medicinal cannabis products manufactured in Australia under a GMP license.
- Medicinal cannabis products listed as export-only, or registered in the Australian Register of Therapeutic Goods (ARTG)
- Extracts of cannabis (or cannabis resin) manufactured under a Narcotic Drugs Act 1967 license, provided they are not in the final dosage form.
The Narcotic Drugs Amendment (Medical Cannabis) Bill 2021, which came into effect on 24 December 2021, introduced a single license model for regulating medicinal cannabis cultivation, production and/or manufacture activities. The licenses granted would be without a fixed expiration date. A medicinal cannabis license can authorize any or all of the following activities for medicinal or scientific purposes:
- cultivation and/or production of cannabis plants, cannabis or cannabis resin
- manufacture of a cannabis drug for one or more permitted supplies.
The new law also introduced permits which are used to manage specific activities on each site such as the volume and type of cannabis (for example, high or low THC).
Recreational Cannabis
In September 2019, the Australian Capital Territory became the first state or territory of Australia to legalize recreational use of cannabis. Since 31 January, 2020, residents aged 18 and over have been allowed to grow two plants per person and possess 50g of dry cannabis or 150g of fresh cannabis. However, sale or other transfers are prohibited, including cannabis seeds.
On June 20, 2023, the Legalize Cannabis Party introduced the “Regulation of Personal Adult Use of Cannabis Bill 2023” in the Victorian, New South Wales and WA state parliaments to legalize the recreational use of cannabis for individuals aged 18 and over. This bill would have allowed possession of up to 50g of cannabis and six plants per household as well as the gifting of up to 50g of cannabis to another adult. The bill also proposed a national cannabis licensing scheme similar to the Canadian model, where cannabis can be sold by both government-run dispensaries and licensed private sellers. The regulator would be responsible for overseeing small-scale commercial production and sales operations, acting as a wholesaler between producers and retailers and setting a wholesale price.
However, in June 2024, this bill was defeated in the WA state and rejected in the Victoria state. Although it has not been formally rejected in New South Wales, debate on the bill has been repeatedly adjourned and there is unlikely to be any momentum for its passage in the near future.
The Legalizing Cannabis Bill 2023 would have legalized recreational cannabis in Australia and established the Cannabis Australia National Agency to regulate the market. However, the bill was not approved.
Australian Cannabis Market Stats
Licensees
As of December 2025, according to Australia’s Department of Health and Aged Care, there were 39 approved medicinal cannabis cultivators and producers, of which 38 are allowed to cultivate and produce, and 22 are allowed to manufacture. There were 23 licensed medicinal cannabis product manufacturers and 36 licensed importers.
Patients and Prescription Statistics
TGA data shows that since its legalization, more than 3.6 million prescriptions for the drug have been issued – either through Authorized Prescribers or through the Special Access Scheme. Prescriptions authorized through the Authorized Prescriber scheme comprise around 77% of this total prescription number.
Prescription numbers were initially small in 2016-2018: a total of around 3,400 over the three-year period. Starting from 2019, year-over-year growth in medical cannabis prescriptions began to accelerate. From 2019 to 2020, they surged by nearly 190% to reach 75,183. Total prescriptions for medicinal cannabis more than tripled to 272,776 in 2021 and then increased by 63% to 445,468 in 2022, with more than half of those prescriptions made through the AP pathway. Annual prescriptions peaked at more than 1 million in 2024 with prescriptions under the AP pathway accounting for more than 80% of the total. In 2025, the market began to show signs of maturing as for the first time prescriptions issued under the AP pathway fell by 15% from the previous year. Meanwhile, SAS-B prescriptions continued to grow by 25%.
Among prescriptions issued through the SAS-B pathway, chronic pain and anxiety were by far the most common conditions authorized for treatment by a medical cannabis product in 2025 (43% and 31% of applications respectively). Other leading conditions approved for treatment included sleep disorder, depression, and post-traumatic stress disorder. Those conditions represented more than 90% of all SAS B prescriptions for medical cannabis.
In 2025, there were 843 medical practitioners approved under the Authorized Prescriber scheme. Data regarding 2025 Authorized Prescriber approvals reveals the medical cannabis dosage forms for which applications were submitted. The most common dosage form was oral liquid (14% of all 2025 applications), followed by pastilles (12%). The pastille dosage form has experienced significant growth in usage with a nearly threefold increase form 2023 to 2025. Dried herb, capsules, and inhalation were other frequently applied for dosage forms.
Sales and Production
North Sydney-based data firm FreshLeaf Analytics estimated the Australian medical cannabis market increased to about $AUD95 million in product sales in 2020, from around $50 million in 2019. According to Penington Institute’s “Cannabis in Australia 2024” report, Australians spent around $234 million on medicinal cannabis products in 2022, reaching $448 million in 2023 and already surpassing $402 million in the first half of 2024.
The report also reveals that there were 2.87 million units of medicinal cannabis sold in the first half of 2024. This is a 71% increase from the 1.68 million units sold in the second half of 2023 and nearly 4 times the units sold in the first half of 2022.
| Medical Cannabis domestically cultivated (kg) | Medical Cannabis imported (kg) | Medical Cannabis exported (kg) | |
| 2021 | 16,700 | 7,306 | 1,426 |
| 2022 | 24,900 | 24,887 | 1,510 |
| 2023 | 26,593 | 44,573 | 2,066 |
| 2024 | 42,400 | 77,406 | 3,312 |
Data from Australia’s Office of Drug Control
In 2022, Australia’s supply of medical cannabis was almost evenly split between domestic cultivation and imports. However, in 2023, imports of medical cannabis soared above domestic cultivation. In 2024, imports of cannabis into Australia reached 77,406 kg – up by nearly 74% from 2023 and 959% from 2021. The top 3 countries importing into Australia were Canada (62,111 kg), South Africa (4,992 kg), Czechia (2,400 kg), and Denmark (2,110 kg).
Although from 2021 to 2023 Germany remained the main destination for Australia’s exported medical cannabis, in 2023 only around 41% of medical cannabis exports went to Germany: this is a large fall considering in 2021, Germany was the destination of 90% of Australian medical cannabis exports. Meanwhile, the amount of cannabis exports to United Kingdom and New Zealand rose to 31% and 28% of total quantity exported in 2023, respectively. In 2024, Australia dramatically ramped up its cannabis exports to New Zealand by 178% and as a result the neighboring country became its largest export market, receiving nearly half of all its exported cannabis supply in that year.
The total amount of cannabis cultivated domestically has also continuously increased and a particularly significant jump occurred from 2023 to 2024 with domestic cannabis production surging by 60% to 42,400 kg.
Australia Cannabis Market Forecast
Cannabis Jobs Australia estimates there will be 50,000 cannabis jobs in Australia by 2028. Australia’s legal cannabis market is forecast to grow from US$600 million in 2024 to US$1.2 billion in 2028, the 5th largest in the world, according to “The Global Cannabis Report: 5th Edition”, suggesting a compound annual growth rate (CAGR) of 18.9%. With this CAGR, the Australian medical cannabis market’s value is estimated at US$714 million in 2025.
The NDSHS 2022-23 Survey found that 41% of people aged 14 and over reported lifetime use of cannabis in the period 2022-23 in Australia; an increase from the 37% reported in 2019. The report also found that 11.5% of people aged 18 and over have used cannabis in the previous 12 months. Support for an Australian adult-use cannabis policy reform is strong with the NDSHS 2022-23 survey finding that over 80% of respondents believe that the possession of cannabis for personal use should not be a criminal offense. Another survey conducted by Roy Morgan (an Australian market research company) from April 2024 to March 2025, revealed that 48% of Australians support adult-use cannabis legalization.
In the Oceania Cannabis Report – produced by London-based advisory group Prohibition Partners – the Australian recreational cannabis market could be worth $3.5b by 2028 if legalized.
Australia’s Parliamentary Budget Office estimates that the legalization of recreational cannabis would bring as much as $28 billion worth of tax revenue (includes cannabis excise tax at 15%, GST, and company tax).
Australian Cannabis Market Infographics
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
Our complete cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
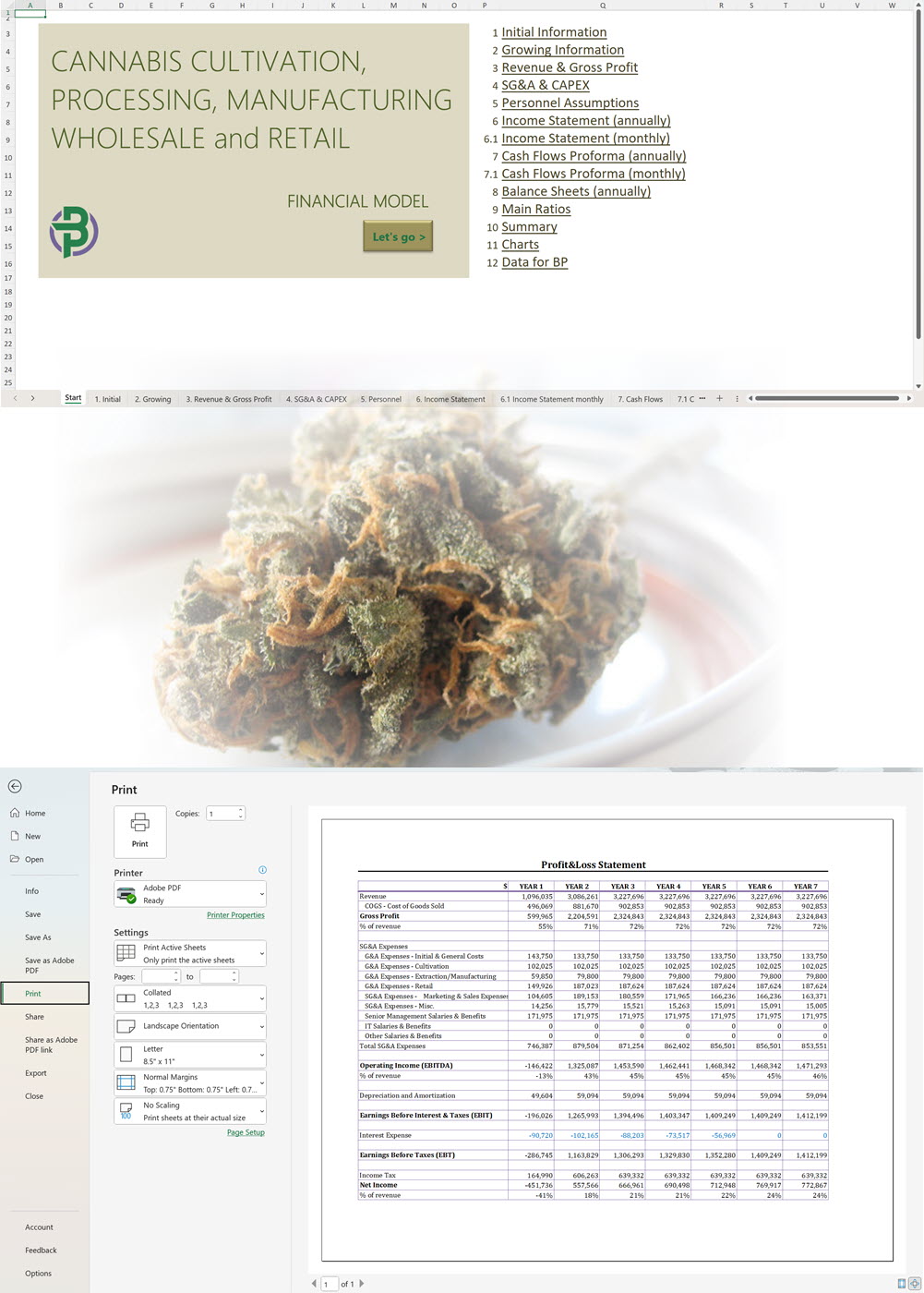
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Cannabis Cultivation and Extraction Business Plan Sample, Australia
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: $75.00 through $350.00 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
$75.00 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
$250.00 Select options This product has multiple variants. The options may be chosen on the product page
Hemp CBD business plan templates are available at hempcbdbusinessplans.com.