Cannabis Legalization
Brazil, the most populated nation in South America, has roughly 214 million people. Since 2014, Brazilians have been organizing medical cannabis associations to democratize access to medical cannabis and raise awareness about its health benefits.
In December 2019, National Sanitary Surveillance Agency (ANVISA) approved RDC 327, legalizing the manufacturing, import and sale of medical cannabis in the country. However, domestic cultivation remained prohibited, meaning companies had to import all raw materials. Although the products are identified as “cannabis-based products”, they are often referred to in other major cannabis markets as hemp products since they must contain mostly CBD and no more than 0.2% THC. However, it is possible for the THC level to be higher in medicinal hemp products that are to be used by patients subject to palliative care or in irreversible or terminal clinical situations.
| Population 2026 (m) | 214.08 | |
| GDP 2026 (US$ t) | 2.29 | |
| Agricultural land (% of land area) | 28.3% | |
| Total spending on health 2025 (US$ b) | 135 | |
| Patients treated with medical cannabis (as of 2025) (est.) | 873,111 | |
| Total of potential cannabis users (est.) | 6.9 million | |
| Source: International Monetary Fund/World Bank Group/International Trade Administration/Kaya Mind | ||
The rule allowed cannabis products without proven efficacy via clinical trials to obtain a “sanitary authorization” from ANVISA which permitted the sale of these products under prescription through pharmacies without needing a special license. Pharmaceutical-quality requirements include:
- Good Manufacturing Practice certification. Until December 2022, ANVISA accepted GMP certifications issued by health agencies of PIC/S countries. From December 2022, only ANVISA certifications are allowed.
- Data demonstrating that the quality of the product remains stable throughout its shelf life in the climatic conditions commonly found in Brazil.
In 2020, some patients began cultivating cannabis at home or through associations, under the protection of Habeas Corpus (HC) which is a constitutional action that aims to guarantee the patient’s freedom when they are threatened or in any way restricted. A total of 42 home or association cultivation HCs were awarded via the justice system from January to July 2020, a 200% increase compared to 2019.
In 2022, through RDC 660, Brazil defined the criteria and procedures for the importation of cannabis-derived products by individuals with a prescription from a legally qualified professional. According to these regulations, nonregistered medical cannabis products can be imported only by individual patients after approval by a doctor and ANVISA.
In October 2022, the Federal Council of Medicine (CFM) published Resolution No. 2324 which introduced restrictions to the therapeutic use of medical cannabis in Brazil but this resolution was then suspended by Resolution No. 2326 in the same month.
On March 14, 2023, the 1st District of the Superior Court of Justice (STJ) suspended all legal actions relating to the cultivation of cannabis for medical, pharmaceutical and industrial purposes until a precedent is set for the matter by the STJ. In June 2024, Brazil decriminalized the possession of up to 40 grams of cannabis or up to six female cannabis plants by an individual.
In November 2024, the Superior Court of Justice issued a ruling that approved the import and cultivation of low-THC cannabis (hemp) in Brazil for the medicinal, pharmaceutical or industrial purposes, and directed ANVISA to create regulations for the production and cultivation of hemp.
On January 28, 2026, ANVISA approved regulatory changes for Brazil’s medical cannabis sector, as was directed by the ruling of the Superior Court of Justice. Five Resolutions from the Collegiate Board (RDC) were approved.
- Domestic Production: The first RDC regulates the domestic cultivation and production of hemp, creating a Special Authorization which companies wishing to produce hemp must obtain.
- Research: The second RDC creates a new regulatory framework for research which allows the use of cannabis plants with THC levels above 0.3% by universities and research institutes for scientific research. This also requires a Special Authorization.
- Associations: The third RDC creates a temporary regulatory framework for non-profit patient associations, allowing small-scale production of hemp. These associations will be monitored to evaluate the sanitary viability of production outside the industrial model as well as the safety and quality of production by these associations. Selection for these associations will occur through a public call for proposals.
- Update Existing Regulations: An update to the regulatory framework for the manufacture and import of medical cannabis products in Brazil. The new regulations permit:
- Compounding of CBD as an active pharmaceutical ingredient (API) in pharmacies.
- New routes of pharmaceutical administration: dermatological, sublingual, buccal, and inhalation use, in addition to the already approved nasal and oral routes.
- Access to medications with a THC concentration greater than 0.2% to be extended to individuals diagnosed with severe debilitating diseases.
- Importation of medical cannabis products.
- Companies to share technical information on cannabis-based medicines, provided it is directed exclusively at healthcare professionals authorized to prescribe and is limited to content approved by ANVISA.
- Update List of Controlled Substances: Cannabis with up to 0.3% THC is removed from the list of prohibited substances.
Sanitary Authorization
In order to manufacture, import, or sell cannabis-derived products, a Sanitary Authorization must be obtained from ANVISA. The permit is valid for five years.
Eligibility:
- Company must have an Operating Authorization and Special Authorization to manufacture medicines, under the terms of RDC No. 16, of April 1, 2014, and its updates. Company must also possess a valid Certificate of Good Manufacturing Practices issued by ANVISA; OR
- Company must have an Operating Authorization and Special Authorization as importers of medicines, under the terms of RDC No. 16, of 2014, and its updates. Company must also possess a valid Certificate of Good Distribution and Storage Practices for medicines.
A company wishing to manufacture or import hemp products for medicinal purpose must submit a request to ANVISA with all necessary documents. Submission must be electronic.
Documents required to be submitted include:
- Petition form for cannabis products, available on ANVISA’s website, duly completed and signed.
- Indication of the number and date of publication of the Operating Authorization and Special Authorization; number of the ANVISA Resolution that grants valid certification of Good Manufacturing Practices for medicines; and number of the ANVISA Resolution that grants valid certification of Good Distribution and Storage Practices for medicines, when applicable.
- Declaration of proof of compliance with Good Manufacturing Practices for Active Pharmaceutical Ingredients (APIs) or Active Pharmaceutical Vegetable Inputs (IFAVs) by active ingredient manufacturer.
- Declaration of proof of compliance with Good Agricultural and Harvesting Practices by the party responsible for cultivating the cannabis plant.
- Declaration issued by the manufacturer of the CBD phytopharmaceutical or extract, or proof of compliance with the legislation in force in the country of origin for the cultivation of the cannabis plant.
- Data on plant species with complete botanical nomenclature and chemotype.
- Technical report regarding the pharmaceutical development of the cannabis product.
- Rationale for the development of the cannabis products.
- In case of IFAV, detailed extraction method for obtaining the cannabis extract.
- Production report, including master formula, batch sizes, list of equipment used, description of the complete production process, in-process controls and flowchart.
- Latest version of the documents containing the quality specifications of the plant drug, the phytopharmaceutical CBD or extract and the finished product, including tests, acceptance criteria and analytical methods.
- Quality control report for raw materials and finished product.
- Document proving compliance with good laboratory practice requirements by the contracted laboratory, if applicable.
- Protocols and reports for the validation of analytical methods and description of methods adopted.
- Protocols and reports for the accelerated and long-term stability studies, for 3 batches of the finished product, and in use, and photostability studies.
- Labeling and packaging layouts.
- Information leaflet layout.
- Declaration of conformity and the model of the Free and Informed Consent Form to be signed by the patient and the prescribing professional, as found in the Annex of the Resolution.
For more detailed information, please see the new regulations approved on January 28, 2026.
Through a Technical Note published in 2023, ANVISA clarified that the import of cannabis flower and plant parts is not allowed – this includes hemp flower and plant parts.
Brazilian Cannabis Market
Sales of medical cannabis in Brazil in 2022 were estimated at $37.1 million, according to Prohibition Partners. Data from ANVISA in 2022 revealed that more than 160,000 patients obtained permits from ANVISA since 2014 through three forms of access – imports, patient associations and pharmacies – with roughly 75% of those prescriptions given in 2022 alone. In 2024, the number of patients who have received treatment with medical cannabis in Brazil surpassed 672,000 which is 56% higher than in 2023. Revenue from medical cannabis reached BRL 852 million: up by 22% from BRL 699 million generated in the previous year.
The Brazilian medical cannabis market in 2025 is estimated at nearly R$ 953 million (approximately US$187 million), according to data from the “Growshops, Headshops and Brands Market 2025” yearbook by the consulting firm Kaya Mind. It is estimated that as of November 2025, there were more than 873,000 medical cannabis patients. Around 40% of them access medical cannabis through imports while 34% obtain cannabis through pharmacies and 26% through patient associations.
According to a survey by Kaya Mind, 79% of cannabis consumers in Brazil use it for feelings of relaxation, with other often mentioned reasons being stress and anxiety relief (64% of respondents) and sleep improvement (45%).
Kaya Mind estimates that around 6.9 million people in Brazil could benefit from medical cannabis as treatment.
In 2023, the Brazilian medical cannabis industry had 516 importers, 276 pharmacies and clinics, 137 patient associations and 27 laboratories. According to the Kaya Mind 2025 Yearbook, the number of patient associations rose by nearly 130% to 315 associations in 2025.
As of January 2026, there were 49 medical cannabis products registered by ANVISA. According to Kaya Mind, in 2025 a total of 68 companies submitted 210 requests for a Sanitary Authorization under RDC 327/19. Kaya Mind estimates that active medical cannabis cultivators have 27 hectares of cultivation land.
Kaya Mind estimates that the total cannabis market in Brazil, including medical, adult-use, and industrial hemp, could have the potential value of up to R$ 26 billion (around US$5 billion) with adult-use accounting for the largest portion of that at R$ 11.7 billion and industrial hemp accounting for R$ 4.5 billion.
Brazil Cannabis Market Infographics
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
Our complete cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
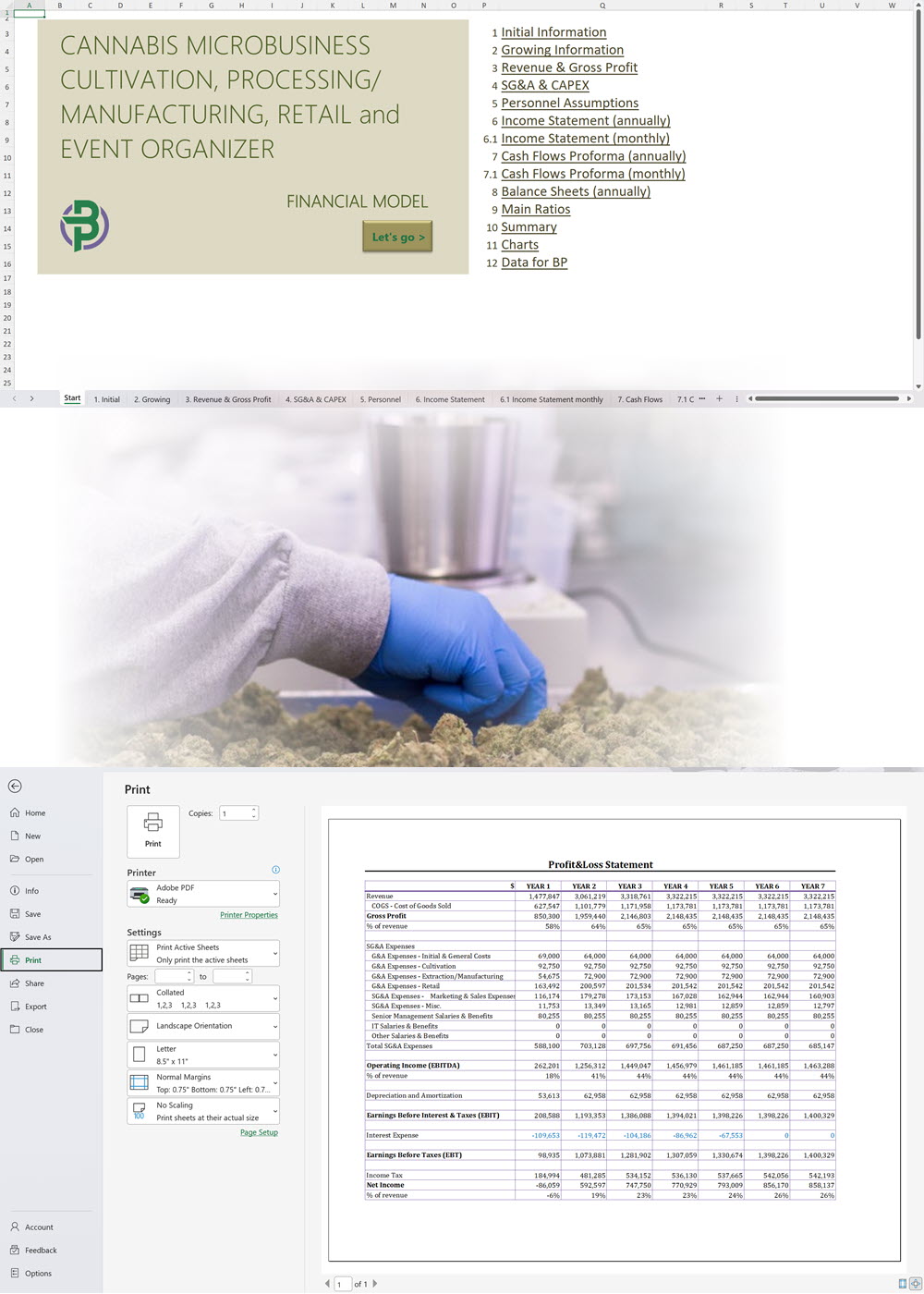
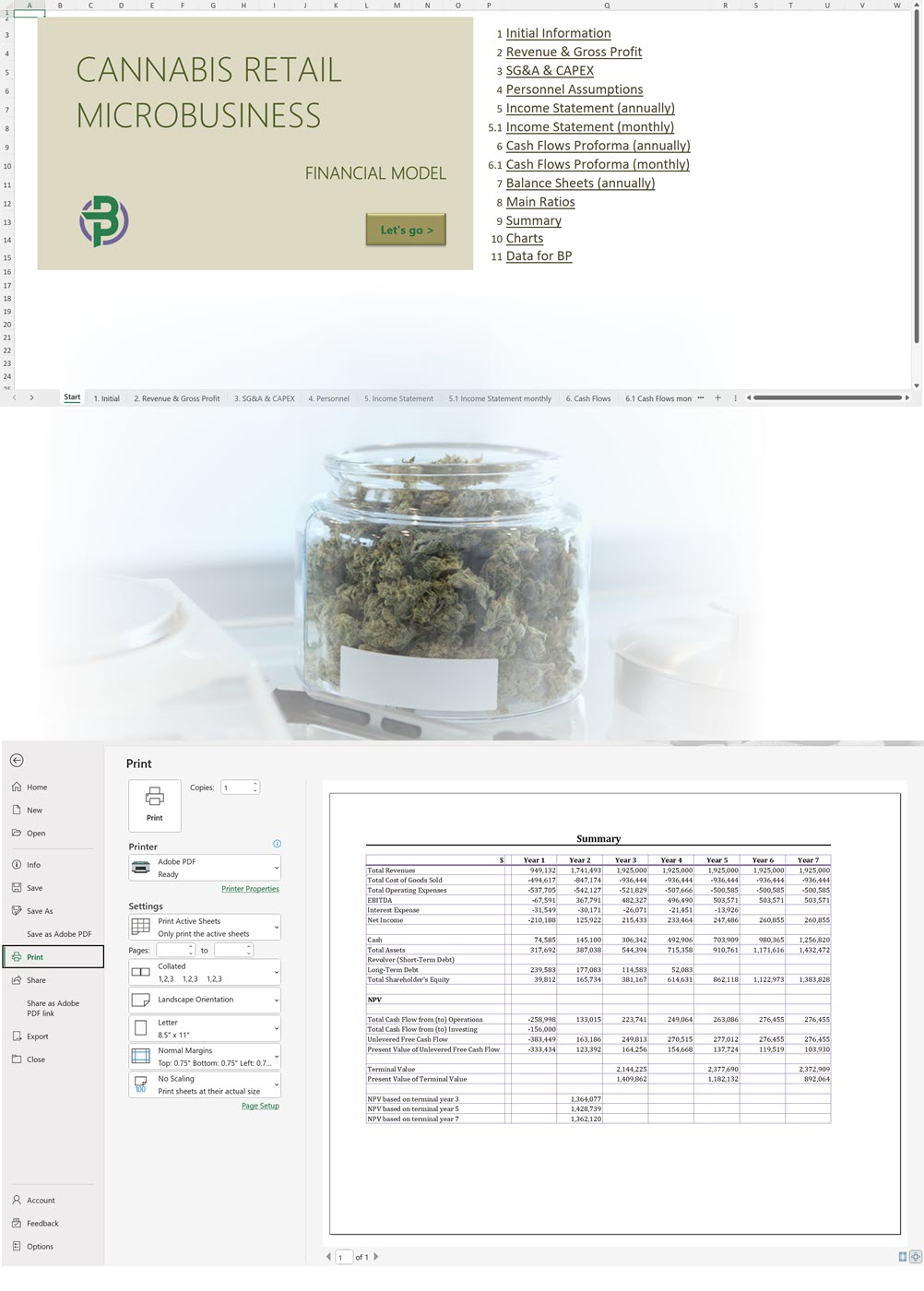
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
CBD Products Manufacturing Business Plan Sample, Brazil
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: $75.00 through $350.00 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
$75.00 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
$250.00 Select options This product has multiple variants. The options may be chosen on the product page
Hemp CBD business plan templates are available at hempcbdbusinessplans.com.