Key Takeaways
- Cannabis law and regulations:
- Permanent legal framework effective January 1, 2026 (Bill L135)
- All licensed physicians may now prescribe
- EU/EØS sourcing permitted for cannabis imports
- Four access pathways: medical cannabis scheme, magistral preparations, approved medicines, and special permits
- Licenses available:
- Manufacturing with cultivation; manufacturing without cultivation; intermediate product manufacturing; special development scheme.
- Market Size & Patient Trends:
- ~1,600 active patients (2024 estimate, +21% vs. 2023)
- Pilot program dominates: 63% of prescriptions in H1 2023 (up from 25% in H1 2018)
- Nearly 20,000 prescriptions issued under the pilot program from H2 2020 to H1 2023
- 90% of pilot program prescriptions are for neuropathic pain
- Industry Landscape:
- 9 licensed bulk/starting product manufacturers (as of 2025)
- 9 licensed intermediate product manufacturers
- 13 products admitted to official medical cannabis product list
- ~9,700+ kg exported in 2024 (primarily to Germany, Australia, UK) and nearly 10,000 kg exported in 2025 to Germany alone
Medical Cannabis
The medical cannabis pilot program in Denmark began in January 2018 and served around 500 unique patients per quarter. Under the program, physicians were allowed to prescribe medical cannabis from a list of five flower and full-spectrum oil products for the following conditions: painful spasms due to multiple sclerosis or spinal cord injury, nausea after chemotherapy, neuropathic pain, although some leeway remains depending on other conditions. 100% of the costs of patients participating in the trial scheme if their condition is terminal are covered by the country’s national insurance agency, and coverage of 50% of the cost is provided in other cases up to DKK 10,000 per year. Patients may also choose to obtain magistral cannabinoid isolates outside of the pilot scheme without reimbursement.
The medical cannabis products allowed for use in the trial can be found on this page.
The scheme was originally conceived as a four-year project, but in May 2021 a majority of Parliament voted to extend the project for another four years, with it now running until at least 2025.
On November 19, 2024, the government proposed that the medical cannabis prescription system should be made permanent after an evaluation of the pilot program showed that medical cannabis was benefitting many patients. On April 24, 2025, the Danish Parliament formally adopted Bill L135, establishing a permanent legislative framework for medical cannabis use, and the new law came into effect on January 1, 2026.
There are four ways that patients can access medicinal cannabis in Denmark:
- Through the medical cannabis scheme.
- As an approved medicine. There are currently two approved medicines: Sativex and Epidyolex.
- Medicines Marinol and Nabilone, which contain synthetically produced cannabinoids, prescribed to patients by a doctor with a special dispensing permit. These medicines can be imported if the Danish Medicines Agency grants a Danish doctor permission to prescribe it to specific patients.
- Medicinal products such as capsules or oils containing ingredients form cannabis prepared in a pharmacy for a specific patient’s needs according to a prescription from a doctor.
All licensed physicians may prescribe medical cannabis based on clinical judgment, without restriction to a pre-defined diagnosis list.
Additionally, the new medical cannabis scheme also permits for cannabis cultivated in EU/EØS countries to be imported for further processing in Denmark (this was previously restricted to domestically grown bulk material for certain product categories). This means that Danish-licensed manufacturers can now source compliant cannabis biomass from other EU countries, potentially lowering production costs and strengthening their supply chain. The scheme also expands pesticide permissions in cannabis cultivation.
Medical Cannabis Licenses in Denmark
All medical cannabis licenses are issued by the Danish Medicines Agency (DMA).
It is important for the applicant to understand the following terms before choosing a permit:
- A cannabis starting product is manufactured from the cannabis plant and could come in various forms such as herbal teas, oils, and capsules.
- A cannabis intermediate product is manufactured by an intermediate product manufacturer from a cannabis primary product with the aim of producing a final cannabis product.
- Cannabis bulk is any processed cannabis product which is not a cannabis starting product or cannabis intermediate product, and which is manufactured for the purpose of further processing or packaging in consumer-ready package sizes either for the purpose of manufacturing a cannabis starting product or for the purpose of export.
- A cannabis product is prepared by a pharmacy according to the prescription presented by a patient.
The following medical cannabis authorizations are available in Denmark:
Allows to produce cannabis starting products with cultivation activity as well as manufacture cannabis bulk and cannabis precursor products. In order to obtain this permit, the applicant must:
- Specify which cannabis products, including product type and product form, are to be manufactured and provide information on the place of manufacture and storage of the cannabis products.
- Have premises and a quality assurance system that meets the requirements of Executive Order No. 1607.
- Have a qualified person available who meets the requirements of Executive Order No. 1607.
- Have a safety responsible person available who meets the requirements of Executive Order No. 1607.
Application fee: DKK 147,909
Annual fee: DKK 60,847
Allows to produce cannabis starting products without cultivation activity as well as manufacture cannabis bulk and cannabis precursor products. In order to obtain this permit, the applicant must:
- Specify which cannabis products, including product type and product form, are to be manufactured and provide information on the place of manufacture and storage of the cannabis products.
- Have premises and a quality assurance system that meets the requirements of Executive Order No. 1607.
- Have a qualified person available who meets the requirements of Executive Order No. 1607.
- Have a safety responsible person available who meets the requirements of Executive Order No. 1607.
Application fee: DKK 142,222
Annual fee: DKK 47,198
Allows the production of cannabis intermediates. In order to obtain the permit, the applicant must:
- Disclose the location of their import of cannabis starting products, receipt of manufactured cannabis starting products, and manufacture and storage of cannabis intermediate products.
- Have premises and a quality assurance system that meets the requirements of Executive Order No. 1608.
- Have a qualified person available who meets the requirements of Executive Order No. 1608.
Application fee: DKK 79,950
Annual fee: DKK 47,198
Additionally, since cannabis is listed in the Executive Order on Euphoriant Substances, an enterprise authorization is required for its cultivation. This cultivation authorization is only issued together with a manufacturing permit under the medical cannabis scheme. When applying for this purpose, the applicant must also submit an application for a permit for the production of bulk cannabis and cannabis precursor products. A detailed guide on how to obtain the cultivation authorization can be found here.
Licensing Process
- Submit a complete application with all the relevant documents to the DMA.
- An inspection will follow, performed by the DMA.
- If the inspection gives satisfactory results, the DMA will issue a certificate authorizing the company to perform the requested activities with medical cannabis and it also provides proof that the company complies with Good Manufacturing Practice for medicines.
Information about the following is required to be included with the application:
- Activities that will be performed with medical cannabis.
- Contractors for outsourced activities, if applicable.
- The Qualified Person & Security Responsible Person of the company.
- The company’s owner or director.
- Address of the proposed location for the medical cannabis establishment.
More information on the application process can be found in the following guides:
Inclusion of Medical Cannabis Products in the Scheme
All cannabis intermediate products must first be admitted to a list published by the DMA before they can be lawfully prescribed by doctors and dispensed by pharmacies in Denmark. Products can either be imported into Denmark or manufactured in Denmark using Danish-grown cannabis. The application forms and instructions on how to submit them can be found on DMA’s official website.
Application fee: DKK 56,059
Annual fee: DKK 40,150
Application processing times for inclusion of cannabis intermediates and related imported cannabis starting products:
Validation – 14 days
Assessment period – 50 days
Application fee: DKK 143,633
Annual fee: DKK 40,150
Application processing times for inclusion of cannabis intermediates and related starting products manufactured in Denmark:
Validation – 14 days
Assessment period – 70 days
Once the product is approved, the company must report information about prices, packaging and delivery capacity.
Import & Export
Since cannabis is listed on List B of the Executive Order on Euphoriant Substances, an import/export certificate is required for all consignments of medical cannabis which cross national borders. Applications for import/export certificates are submitted to the DMA through NDS Web (National Drug Control System Web).
Fees are collected for the issuance of import certificates. The fee is DKK 1,625 per certificate.
Special Development Scheme
On January 1, 2018, Denmark introduced a special development scheme under which companies can apply for an authorization from the DMA to cultivate and handle cannabis for the purpose of producing medical cannabis. Though the authorization does allow to grow and develop medical cannabis, it does not allow the company to supply medical cannabis products for patient use in Denmark or export them; for this, a cannabis bulk manufacturing permit is required. The DMA official website states that the purpose of an authorization under the development scheme is to give companies “an opportunity to build competencies and develop methods and products within medical cannabis for use in the trial scheme”. Later, the company can apply for a cannabis bulk manufacturing permit in order to be able to supply its products for patient use and export.
Application fee: DKK 49,594
Annual fee: DKK 15,102
To obtain the authorization, the applicant must submit a complete application and a project description to the DMA and the DMA must also carry out an inspection. The project description should demonstrate how the applicant’s proposed project will contribute to the development of cannabis products that can be included in the medical cannabis trial scheme or used in pharmaceutical production.
The application must contain the following information:
- The type of activity the applicant wants to perform relating to medical cannabis. Possible activities include import, export, receipt of cannabis, cultivation, possession and preparation of products and solutions.
- The addresses of the locations where cultivation and/or processing will take place.
- If the applicant intends to cultivate cannabis, the cultivation area must be stated in hectares.
- Information about the person responsible for security and about the company’s owner or director. Both must sign their consent that the DMA can access information about personal circumstances from the National Police.
The project description must provide information on the following topics:
- Quality assurance.
- Personnel and training.
- Buildings and facilities.
- Equipment.
- Seeds and propagating material.
- Cultivation process.
- Harvesting process.
- Primary processing which includes washing, trimming and drying.
- Packaging of the harvested cannabis.
- Storage of cannabis product.
- Timetable for cultivation, harvesting and primary processing as well as when cannabis products will be included in the medical cannabis trial scheme.
- Destruction of cannabis procedure.
- Special safety measures relating to the cultivation and processing of cannabis.
- Production of medicinal cannabis.
The DMA has a guide on how to apply for the authorization as well as a detailed guide on what information the project description should contain.
Cannabis Market in Denmark
According to a presentation from the DMA on October 9, 2025, there are 9 companies holding a permit to produce cannabis bulk and starting material, and 9 companies holding a permit to manufacture intermediate cannabis products.
As of April 2026, there were 13 cannabis intermediate products in DMA’s list of admitted products.
According to a November 2024 Trial Evaluation Report, there were nearly 3,000 medical cannabis patients in 2018 across all four access pathways. Patient numbers peaked at 3,569 in 2019 before declining to just below 3,000 in 2020 and falling further to 2,494 in 2022. Prescriptions volumes, however, followed a different trajectory: after rising 33.5% from 2018 to 2019, they continued growing to reach 13,432 prescriptions in 2022. This growth was driven almost entirely by the medical cannabis pilot program, while magistral preparations, special permits, and approved medicines all saw prescription declines over the same period.
Despite a 55% decline in unique pilot program patients – from 1,705 in 2019 to just 768 in 2022 – annual prescriptions more than doubled from 2,951 to 6,469. Prescriptions per patient surged from approximately 2.4 to 8.5, suggesting the pilot program consolidated around a smaller group of highly adherent, long-term users who found clinical benefit. In 2022, pilot program prescriptions surpassed magistral preparations for the first time; by H1 2023, they accounted for 63% of all medical cannabis prescriptions, up from just 25% in H1 2018. Nearly 20,000 pilot program prescriptions were issued from H2 2020 to H1 2023.
A rebound emerged in H1 2023: pilot program patients doubled to 962 year-over-year, with prescriptions rising 84.5%. It is estimated that pilot program participation reached ~1,326 patients in the full-year 2023 and rose further by 21% to 1,605 in 2024. With the permanent framework now permitting all licensed physicians to prescribe medical cannabis based on clinical judgment – without restriction to a pre-defined diagnosis list – patient access is likely to expand further.
Products from Aurora’s facilities hit the German shelves in February 2021, marking the country’s first international export. In all of 2021, 3,424kg of medical cannabis were exported from Denmark. This rose by 55% to 5,313kg in 2022 and an estimated 4,501kg in 2023 (through November 30, 2023) according to the November 2024 Trial Evaluation Report. In 2024, based on data provided by Germany, UK, and Australia on their medical cannabis imports, Denmark exported 9,749kg of medical cannabis to those countries with the majority of the amount (~76%) going to Germany. In 2025, nearly 10,000kg of medical cannabis was exported from Denmark to Germany.
In Denmark, the majority of the market share is in single substance extracts, both THC and CBD, with flowers accounting for only 12% of the total unlicensed market. Around 90% of pilot program prescriptions are for neuropathic pain.
According to Prohibition Partners, Denmark’s medical cannabis market was valued at more than €3.5 million in 2025.
Source: cannabusinessplans.eu.
Denmark Cannabis Market Infographics
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
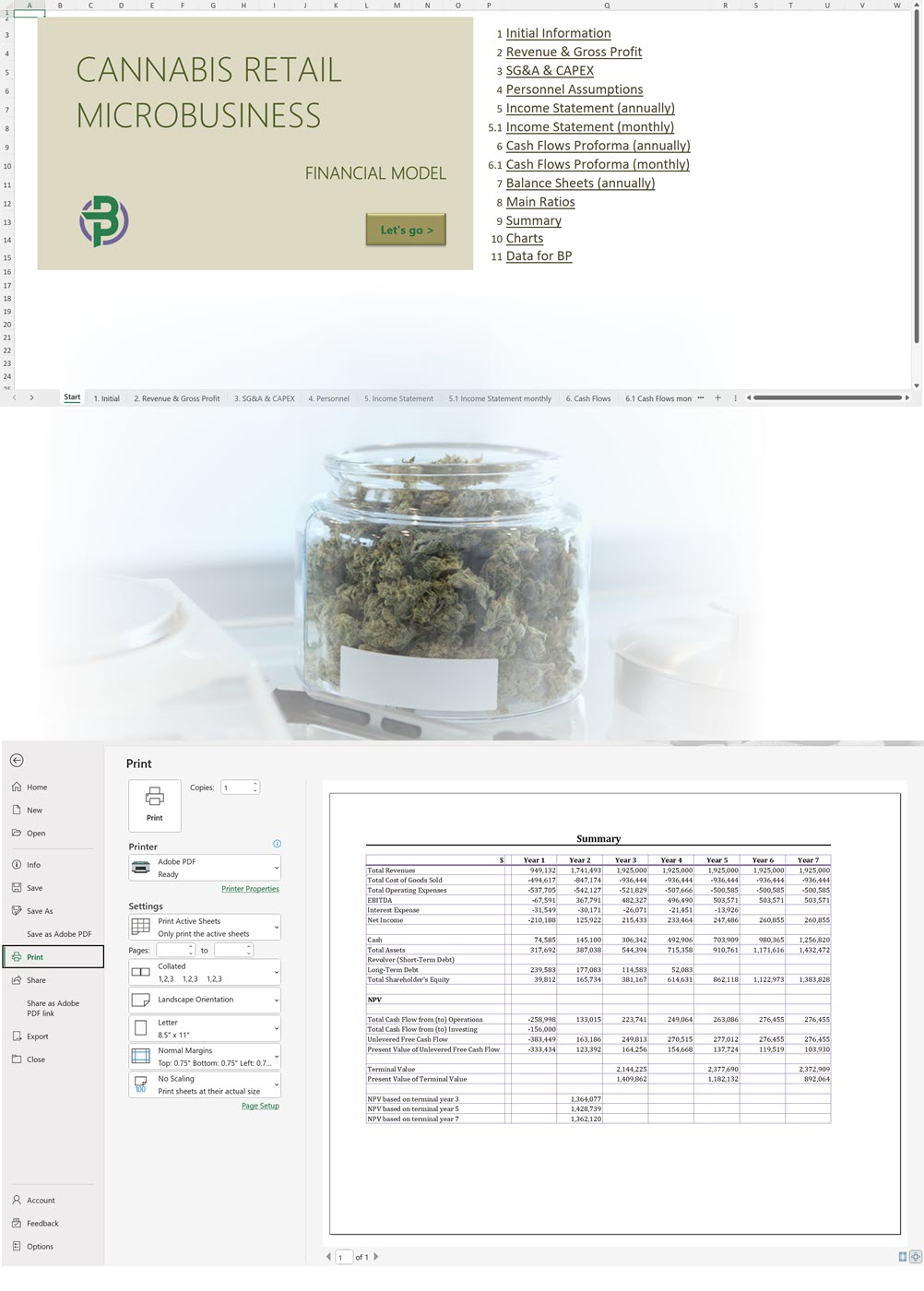
Our complete cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
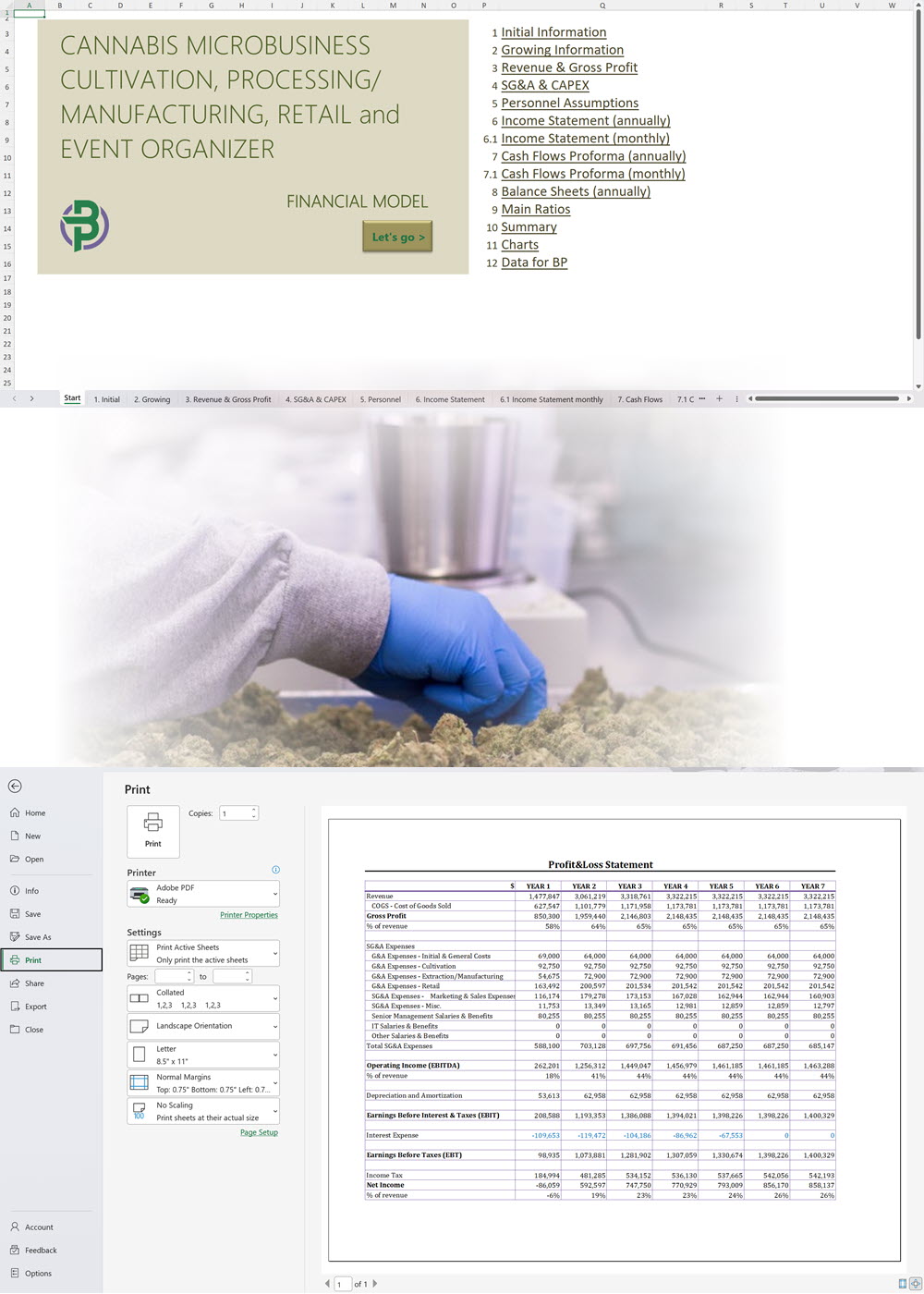
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Cannabis Cultivation, Extraction and Manufacturing Pitch Deck Sample, Denmark
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: $75.00 through $350.00 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
$75.00 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
$250.00 Select options This product has multiple variants. The options may be chosen on the product page
Cannabis business plan templates for European countries can be found at cannabusinessplans.eu.
We also offer hemp/CBD business plan templates at hempcbdbusinessplans.com.